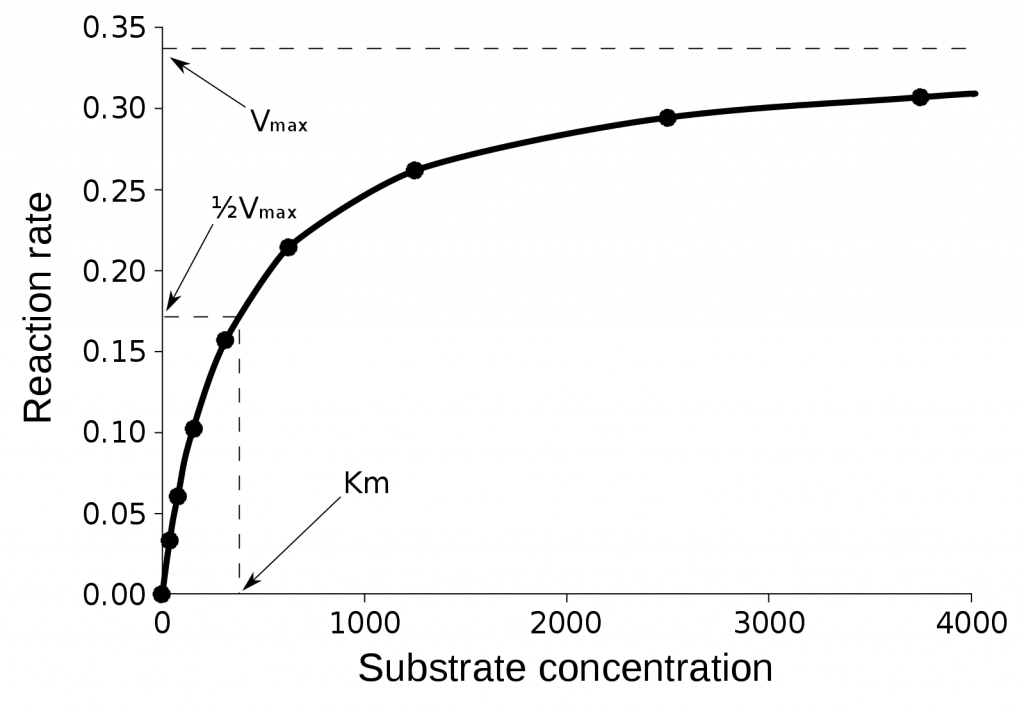
Michaelis Menten equation is used to study the kinetics of enzymatic reaction. Usually, the rate of reaction (a.k.a. velocity) is measured at several different substrate concentration values.
Below there is a succinct explanation of Michaelis Menten equation.
http://varunyilunusabowebsite.weebly.com/uploads/3/7/5/4/37541075/michaelis_menten_equation.pdf